In Switzerland, solid organ donor evaluation and organ allocation have been well organized at the level of six transplant centers since 1985. Each transplant program collected its own data and no monitoring or auditing was performed. No country-wide structure existed to systematically monitor transplant outcomes and to coordinate multicenter studies for the post-transplant process.
In 2006, several Swiss investigators from different disciplines decided to launch a prospective multicenter cohort project, the Swiss Transplant Cohort Study (STCS), aiming at a nationwide comprehensive and structured data collection in all solid organ transplant (SOT) recipients. After a two-year set-up period the STCS started patient enrolment in May 2008.
In a parallel development, a new transplantation law was enforced in 2007, requiring a mandatory lifelong follow up of all transplanted patients in Switzerland. In a collaborative effort with the Federal Office of Public Health, the cohort ensures compliance with the requirements of the law.
Other national and international transplant registries have previously shown their value by generating a large body of knowledge in transplantation medicine. Examples are the Scientific Registry of Transplant Recipients (SRTR) in the US, the Heidelberg-based Collaborative Transplant Study (CTS) in Europe, the Australia and New Zealand Dialysis and Transplant (ANZDATA) registry, as well as the Spanish Resitra cohort. These large registries focus on specific biomedical factors but are often limited in regard to the integration of psychosocial and behavioral determinants, infectious disease occurrence, immunologic determinants and a variety of long-term outcome. Moreover, most of these registries are based on willingness of the centers to contribute patient data.
One of the aims of the STCS is to provide a prolonged follow-up and embracing a comprehensive bio-psychosocial perspective in its data collection providing a unique instrument for comparative effectiveness research.
The main objectives of the STCS are to:
The local transplant coordinators have full access to the Swiss organ procurement system (SOAS) and are informed about all listings and transplantations. They work in close collaboration with the STCS local data managers and provide information about enlisted patients and all recent transplantations performed at their transplant center.
Local transplant physicians and data managers prospectively collect the requested data. Written information about the STCS and informed consent is obtained from listed patients prior to transplantation. All data are entered into patient- and organ-specific online case report forms (CRF) and all data are electronically transferred to a central database system.
Standard operating procedures (SOPs) establish the working standards, address legal aspects of consent handling, bio-sampling and on updating of the STCS infrastructure.
The core data sets, that are key for reporting and research, are continuously subjected to quality checks throughout Switzerland and incongruences are corrected in collaboration with the transplant centers and the FOPH. Furthermore, the FOPH and the STCS work together toward data consistency and cleaning at regular intervals, as required by the Swiss Transplantation Ordinance.
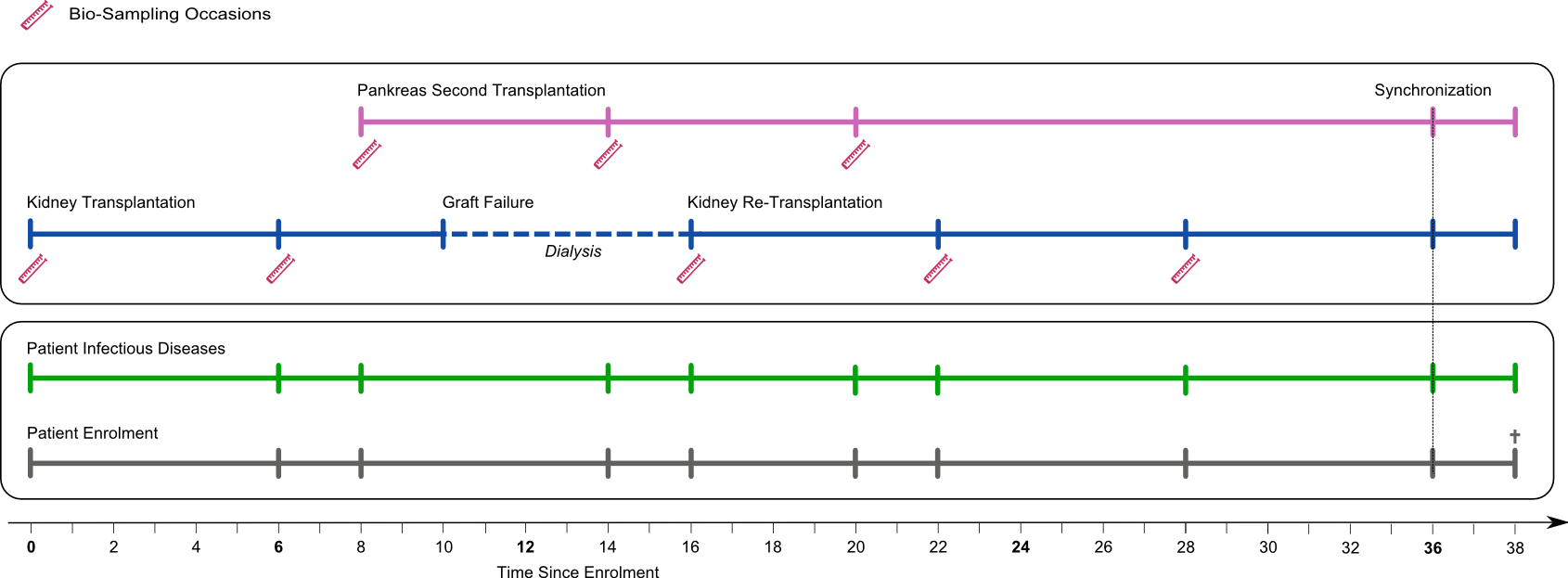
The STCS is a multi-centre cohort study designed as a flexible and dynamic patient-case system. It allows capturing all transplant scenarios of any complexity and distinguishing data that accrue in relation to the patient from data related to the transplanted organ(s). Beside the medical data, specific focus is directed at infectious diseases, bio-banking, psychosocial and behavioral factors.
The data collection process (patient timeline) starts with the transplantation or the moment when the islets are injected into the recipient. After a baseline assessment that contains all patients- and transplant-relevant data, all patients are mandatorily followed in their respective transplant centers.
STCS follow-up assessments take place at six and twelve months and yearly thereafter. Data are collected longitudinally and are thus updated at each visit. The only exception is past medical history data, which are collected at baseline only.
In case of a second or re-transplantation, the STCS performs three extra assessments of the new case at baseline, six, and twelve month. The patient data is updated in regard to the new case. After completion of these three extra visits, the schedule is synchronized to the initial patient visit schedule (patient timeline). Biological samples are collected to the case at baseline and at the six and twelve-month visits.
The psychosocial assessment is performed before the transplantation, at 6-, and 12 months and yearly thereafter along the patient timeline. The psychosocial assessment is a self-report questionnaire that integrates validated instruments as well as items from other cohort studies or nationwide surveys.
A patient follow-up ends with death or definitive drop out at 38 months after the first enrollment. Non-fatal graft failures do not truncate a patient's follow up.
All data, collected by local transplant physicians and data managers, are entered into patient- and organ-specific online case report forms (CRF) and electronically transferred to a central database system. Working standards are established in the STCS-standard operating procedures (SOPs).
Figure: Organization of the STCS patient-case system based on a hypothetical complex transplantation scenario